A single contaminated batch of face cream recalled from shelves costs a mid-size cosmetics brand an average of $47,000 in direct losses — before you count the regulatory fines, the destroyed inventory, and the 6-month brand recovery cost.
We have seen it happen. Twice. Both times, the brands were running quality checks on paper checklists and Excel sheets.
If you are manufacturing cosmetics and you are not running a structured Odoo quality control system inside your manufacturing workflow, you are gambling with your entire product line every single day.
The Compliance Nightmare Your QA Manager Hides From You
Here is what actually happens on a production floor without proper QC automation: your QA manager creates a Word document template for inspection forms, prints 40 copies every Monday morning, and then someone files them in a binder that nobody looks at until there is an audit.
The FDA's Modernization of Cosmetics Regulation Act (MoCRA) now gives regulators the authority to walk into your facility and demand full batch traceability records on demand. Under ISO 22716 GMP guidelines, you are required to document every raw material lot used in each production batch, every in-process check, and every deviation — with timestamps and signatures.
The $8,400 Audit Scramble
When that inspector shows up and asks for the microbiological test results from batch #L24-0089 produced eight months ago, what does your team do? Dig through a binder? Check the shared Google Drive folder?
We watched a $3.2M UAE cosmetics brand spend 37 hours preparing documentation for a single FDA audit — documentation that should have taken 4 minutes to pull from a properly configured ERP system.
That is $8,400 in lost labor hours for one audit.
Multiply by 3-4 audits per year: $33,600 annually just scrambling for paperwork.
Why Your Current QC Setup Is a Ticking Recall Clock
We know what most cosmetics brands are doing. You have a checklist in Google Sheets. Your QA supervisor manually enters viscosity readings, pH levels, and microbial test results after the batch is already packaged. By the time someone notices that the pH of your vitamin C serum batch drifted from 3.2 to 4.1 — which completely destabilizes the active ingredient — it is already in 1,200 units sitting in your warehouse.
The False Solution: Hiring More QA Staff
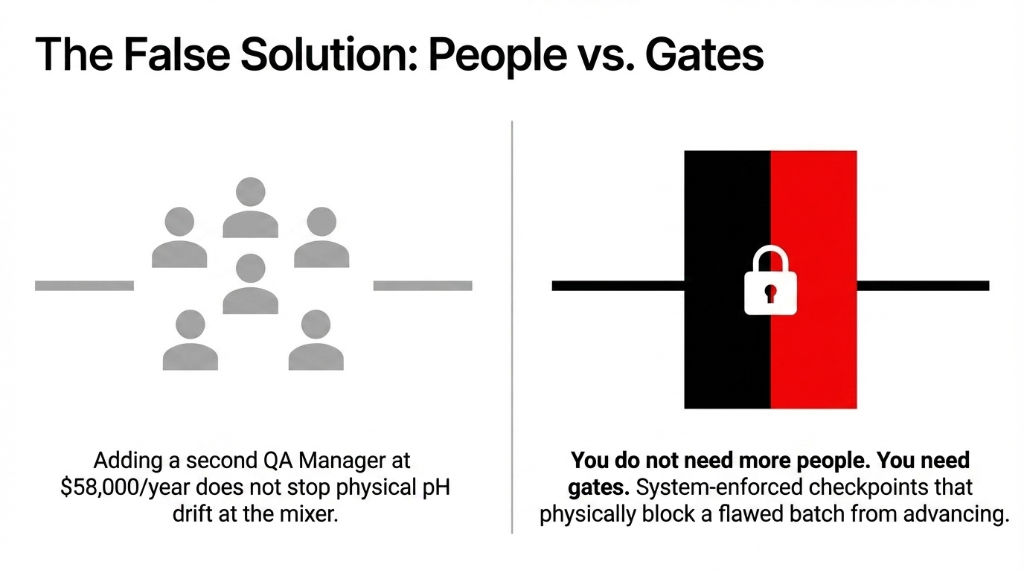
The controversial opinion nobody will say out loud: hiring more QA staff is not the solution. We see founders do this constantly — they add a second QA manager at $58,000/year when the real problem is that there is no system forcing the check to happen before the batch moves to the next production stage.
You do not need more people. You need gates. System-enforced checkpoints that physically block a flawed batch from advancing.
Odoo's quality control module creates exactly those gates — automated checkpoints that physically block a manufacturing work order from advancing until the inspection passes. No more discovering pH drift at packaging. The check happens at the mixer, with a mandatory pass/fail recorded directly in the system, timestamped, and attached to that lot number permanently.
How Odoo QC Actually Works Inside a Cosmetics Plant
Here is the operational reality of how we configure Odoo quality control for cosmetics manufacturing — not the sales pitch version.
Quality Control Points (QCPs) are the engine. You configure them in Odoo to trigger at specific operations: incoming raw material receipt, compounding/blending, filling, and final packaging. For a typical skincare brand, we set up checkpoints at a minimum of 4 production stages:
The 4 QC Gates for Cosmetics Manufacturing
Gate 1: Incoming Raw Material Inspection
Supplier lot documentation verified, CoA (Certificate of Analysis) matched against your internal specs before any material enters production. Failed lots are auto-quarantined.
Gate 2: Pre-Blend Check
pH range validation, viscosity baseline, temperature confirmation. This is where pH drift from 3.2 to 4.1 gets caught — at the mixer, not at packaging.
Gate 3: Post-Fill Inspection
Fill weight accuracy (usually ±0.5g tolerance), cap torque, visual seal check. Catches underfilling and seal failures before boxing.
Gate 4: Pre-Shipment Final QC
Microbiological testing cross-reference, label accuracy against the regulatory specification sheet. Nothing ships until this passes.
Each QCP generates a digital quality check record in Odoo. The check record captures the inspector's name, the timestamp, the test result, and a pass/fail status — all linked to the specific manufacturing order and lot number. If a check fails, Odoo automatically flags the batch as quarantined and routes it to a scrap or rework decision workflow. The batch cannot move to shipping. Period.
This is the gate your team currently does not have. No manual "yellow sticker" system. No risk that your warehouse operator accidentally grabs the failed batch because the sticker fell off. Odoo physically blocks it in the system.
Batch Traceability Is Your Recall Insurance Policy
In our implementations across the UK and US for cosmetics brands doing $1M-$12M ARR, the single highest-ROI feature is lot and serial number traceability inside Odoo's manufacturing module.
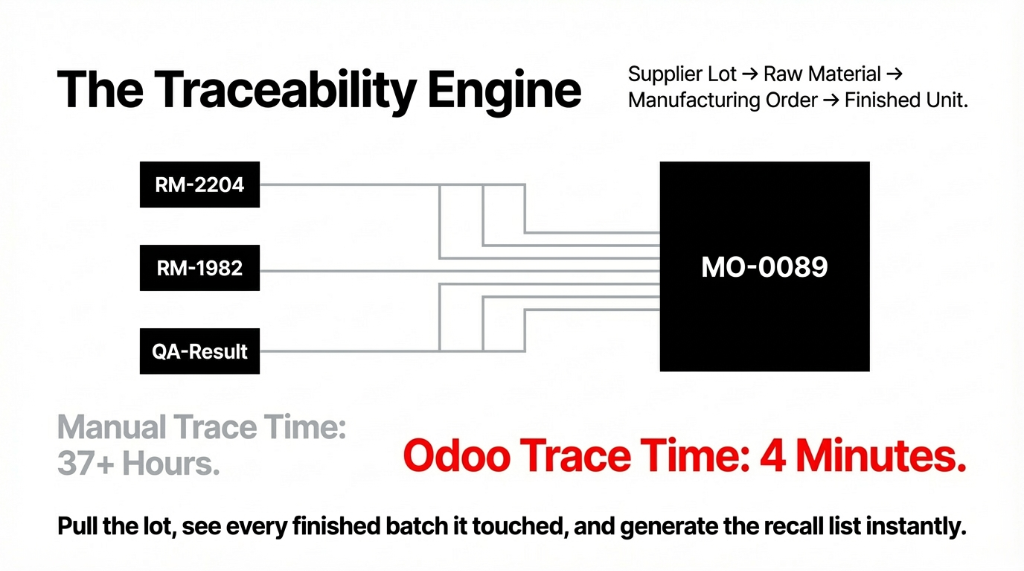
Every raw material that enters your facility gets assigned a lot number tied to the supplier, the incoming date, the QC test results, and the CoA. When that ingredient gets consumed in a production batch, Odoo records exactly how much of Lot #RM-2204 went into Manufacturing Order #MO-0089. When MO-0089 produces 800 units of finished product, every single unit carries a traceable link back to every raw material lot used.
The Insider Detail That Matters
When the FDA asks you to trace a complaint back to source — or worse, when you need to initiate a voluntary recall — the entire trace takes 4 minutes in Odoo versus 37+ hours in a manual system.
You pull the lot, you see every finished batch it touched, you see every customer order those batches shipped to, and you generate the recall list. Done.
ISO 22716 requires exactly this level of traceability. Odoo delivers it out of the box — but only if you configure it properly from day one. (This is where most generic Odoo implementations fail cosmetics brands — they set up the module for generic manufacturing without mapping it to GMP-specific workflows.)
The Numbers You Should Actually Expect
We are not going to promise you vague "efficiency gains." Here is what we have measured across our cosmetics manufacturing implementations:
Verified Results From Odoo QC Implementations
Audit Preparation Time
From 37 hours per audit → 3.5 hours
Documentation retrieval fully automated
Batch Release Cycle Time
From 2.3 days average → 11 hours
QC checks embedded in workflow, no paper sign-offs
Rework Rate Reduction
31% reduction in rework batches within 90 days
Upstream QC gates catch deviations before compounding errors stack
Regulatory Non-Conformance Avoided
UAE client avoided a $28,000 potential fine
First post-implementation audit — every document timestamped and traceable
Inventory integration: When a raw material lot fails incoming QC, it is automatically quarantined in a separate warehouse location in the system and cannot be picked for production. No manual "yellow sticker" system. No risk that your warehouse operator accidentally grabs the failed batch because the sticker fell off.
What a Real Implementation Looks Like
We get asked this constantly: how long does it actually take? A properly scoped Odoo QC implementation for a cosmetics manufacturer with 20-80 SKUs takes 6 to 9 weeks. Not 6 months. Not "it depends."
| Phase | What Happens | What You Get |
|---|---|---|
| Week 1-2 | QC point mapping — document every inspection step and map to Odoo QCP configurations | Complete QC blueprint for your process |
| Week 3-4 | Lot traceability setup — raw material and finished goods lot structures configured, BOM traceability chains built | Full supplier-to-product lot linkage |
| Week 5-6 | User acceptance testing — QA team runs 3 full mock production cycles, fail/quarantine routing tuned | Battle-tested gates before go-live |
| Week 7-9 | Go-live + 2-week hypercare support — on-call during first live batches | Live system with safety net |
Day 1 vs. Day 30
Day 1: QA managers stop re-entering test results from paper forms into Excel. That alone saves 4.5 hours per week per QA manager.
Day 30: Your production supervisors start trusting the system to block batch advancement instead of relying on verbal approvals. That is when the real quality improvement kicks in.
Everyone says buy NetSuite for this. Do not. It burns $500k and takes 12 months before your first batch record is digital. Odoo does it in 6-9 weeks and your QA team is live — not waiting for a consultant to finish "requirements gathering."
We have implemented this exact system for cosmetics brands doing $800K to $14M ARR — across the US, UAE, and UK. The ROI is not theoretical. It shows up in your first post-implementation audit. And it connects directly to your D2C fulfillment — so quality holds flow through to shipping blocks automatically.
Frequently Asked Questions
Does Odoo quality control support ISO 22716 GMP requirements for cosmetics?
Yes. Odoo's QC module covers all core ISO 22716 requirements — batch records, in-process check documentation, raw material traceability, and audit trails with timestamps. With proper configuration by a specialist like Braincuber, it maps directly to GMP documentation standards without requiring third-party add-ons.
Can Odoo handle microbiological test results and stability testing records?
Odoo's quality module supports custom test types, so you can configure it to record microbial count results, stability test readings, and shelf-life data — all linked to the batch lot number. Results are stored permanently and are searchable in seconds during audits.
What happens if a quality check fails mid-production in Odoo?
The manufacturing work order is automatically blocked from advancing to the next stage. The failed batch is flagged and routed to a quarantine location in inventory. A notification is triggered to the QA manager. No batch moves forward without a deliberate pass decision or approved deviation record.
How does Odoo connect QC data to supplier performance tracking?
Every incoming raw material QC result is logged against the supplier's lot and purchase order. Over time, Odoo builds a rejection history per supplier — if Supplier X fails incoming QC on 3 of 7 deliveries, that pattern is visible in reports, giving your procurement team hard data for renegotiation or supplier replacement.
Is Odoo quality control expensive to implement for a small cosmetics brand?
A focused Odoo QC implementation for a small cosmetics manufacturer (10-50 SKUs) costs significantly less than one product recall or one missed compliance audit. Braincuber's scoped implementations start at a fraction of what NetSuite charges for equivalent functionality — and go live in 6-9 weeks, not 12 months.
Pull Your Last Batch Record. Time How Long It Takes.
If finding a single batch's full traceability chain takes more than 5 minutes, you have a compliance gap that will cost you the next time an inspector walks in. Book our free 15-Minute Operations Audit — we will identify your biggest QC exposure in the first call.
Do not let a single failed batch cost you $47,000 and three months of brand recovery.

